Thermodynamics
Objective for this Unit: Apply thermodynamic equations to real life examples.
Objective Summary:
We did practice problems for the different worksheets we were handed and had an isolation lab to see how high we could heat up the box using our own isolation material that we came up with.
We did practice problems for the different worksheets we were handed and had an isolation lab to see how high we could heat up the box using our own isolation material that we came up with.
Definition of Thermodynamics:
The study of the effects of work, heat, flow, and energy on a system.
The study of the effects of work, heat, flow, and energy on a system.
Variables List:
Q= Energy transferred
m= mass of material (kilograms)
c= specific heat capacity of material (Joules/ kilograms Celsius)
ΔT= Change in temperature
P= rate of energy transfer (watts)
k= thermal conductivity
A= area of thermal conductivity
Δt= Change in time
L= thickness of material
Q= Energy transferred
m= mass of material (kilograms)
c= specific heat capacity of material (Joules/ kilograms Celsius)
ΔT= Change in temperature
P= rate of energy transfer (watts)
k= thermal conductivity
A= area of thermal conductivity
Δt= Change in time
L= thickness of material
Equations List:
Energy Transfer:
Q=m*c*ΔT
-Used to figure out how much energy is lost by a hotter object and gained
by the colder object.
Rate of Energy Transfer:
P= kA*ΔT/L
-Used to find the average rate at which heat is released or gained by and object
(Conduction, convection, and radiation).
P= Q/ΔT
-Used when you need to find Q when you already know the rate of energy transfer.
Thermal Conductivity:
k=PL/A*ΔT
-Used to find out how good your material can conduct energy. Dividing the energy
transfer rate by the area and change in time, you get the conductivity of the
equation.
U-Value:
P/AΔT:
-Used to measure the material's ability to conduct heat.
R-Value:
R= 1/U
-Used to measure the material's ability to resist heat.
Insulation Lab
Objective: Team will find the best isolation material to keep heat inside a heat box.
Materials:
- Heat Box
- LoggerPro Software
- Insulation Material
- Tape
- Ruler
- 2 Stainless steel Temperature Probes (for heat box)
Design:
Our team's design was to make something that would keep heat inside so we compacted a variety of materials.
It has denim, cotton, aluminum foil, and cardboard. The only constraint to designing this was to make sure it didn't go over 1 inch in thickness. Our design stayed below that at 0.8 in.
Our team's design was to make something that would keep heat inside so we compacted a variety of materials.
It has denim, cotton, aluminum foil, and cardboard. The only constraint to designing this was to make sure it didn't go over 1 inch in thickness. Our design stayed below that at 0.8 in.
This is a close up of the denim and cotton.
Our goal for this design was for the heat to be neutral in the case that both temperature probes would be hot. This is done because aluminum has two different uses for energy. It absorbs it and reflect it. It reflects it back into the other insolation making the box hot and absorbs it making the top side of the box hot as well. The cardboard was also used to keep the materials together and worked out to be a good material to keep heat inside.
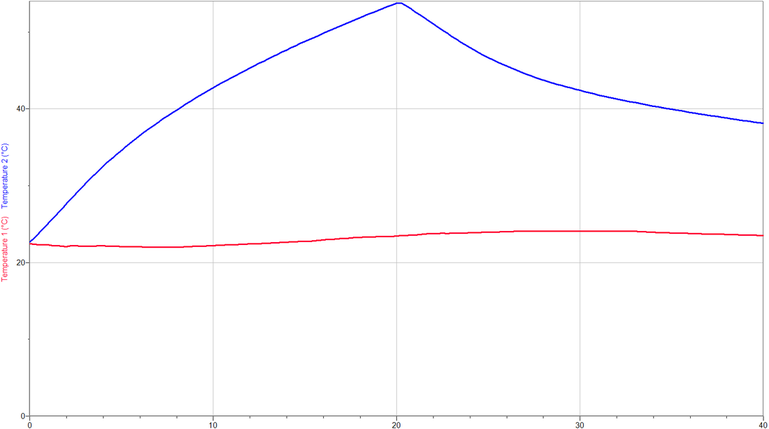
Temperature Data
Data for Box:
Length: .144 m.
Width: .143 m.
Height: .173 m.
Volume: .003562416 m^3
Length: .144 m.
Width: .143 m.
Height: .173 m.
Volume: .003562416 m^3
|
Energy Gained by Air:
Q=130.68 Joules Energy Lost by Air: Q=-63.72 Joules Net Energy Retained: Qnet= 66.96 |
Heat Source: 25 Watt Light Bulb
Initial Internal Temp.: 22.5 degrees Celsius Maximum in ternal temperature: 55.8 Celsius Final internal temperature: 38.1 Celsius Inital room temperature: 22.7 Celsius Maximum room temperatue: 23.7 Celsius Final room temperature: 23.5 Celsius Heating time(s): 20 min Cooling time(s): 20 min |
Conclusion:
This unit had proved to teach me more in depth of how thermodynamics work. It branches all the way from calculating it to measuring it with a probe. The insulation lab was fun because we got to choose what our insulation materials would be used so it contributed to creativity. Our design worked out well because of the aluminum used to reflect the energy. Overall, this was one of the best labs next to the simple machines lab.
This unit had proved to teach me more in depth of how thermodynamics work. It branches all the way from calculating it to measuring it with a probe. The insulation lab was fun because we got to choose what our insulation materials would be used so it contributed to creativity. Our design worked out well because of the aluminum used to reflect the energy. Overall, this was one of the best labs next to the simple machines lab.